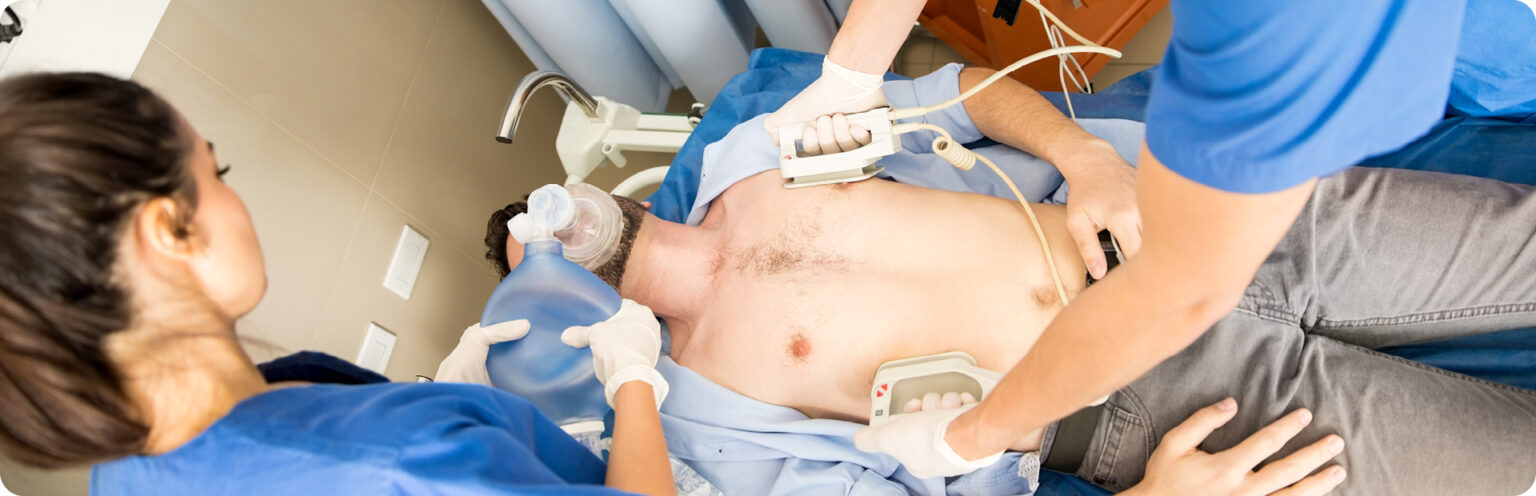
There are four possible electrocardiographic rhythms in cardiac arrest: ventricular fibrillation (VF), pulseless ventricular tachycardia (VT), pulseless electrical activity (PEA), and asystole. VF is a disorganized electrical activity, while a pulseless VT produces an organized electrical activity. Both of these electrical states are incapable of producing sufficient blood flow. PEA is an organized electric rhythm that is characterized by an absence or insufficient mechanical ventricular activity to produce a detectable pulse. Asystole is an absence of ventricular electrical activity with or without atrial electric activity. The chances of survival decrease as the delay of defibrillation in VF and pulseless VT increases.1-2 Therefore, hospitals should develop a plan to reduce the interval between arrest and rapid defibrillation.3 Outcomes in defibrillation are also optimized if the interruptions between chest compressions are minimized. This has resulted in a significant improvement in the rate of survival to hospital discharge.4-10
Conversely, it is important to note that other advanced cardiac life support (ACLS) therapies, such as medical interventions and advanced airways, may improve the likelihood of return of spontaneous circulation (ROSC), but have not shown significant improvement in the rate of survival to hospital discharge.11-17 The key to rhythm-based management is that interruptions in cardiopulmonary resuscitation (CPR) should be as short as possible, giving way only to assess the rhythm, shock VF or pulseless VT if indicated, perform a pulse check when an organized rhythm is achieved or to place an airway.
The arrest rhythm frequently changes during the course of resuscitation, and management should be modified to the appropriate rhythm-based strategy. For example, the health care providers should be prepared to immediately defibrillate a cardiac arrest victim in asystole or PEA who converts into VF or pulseless VT during a rhythm check. The following describes the management of the cardiac arrest victim according to rhythm.
- VF or pulseless VT – a rhythm check on an AED after 2 minutes of CPR in a victim with VF or pulseless VT will prompt spontaneous charging of the device and prompt the health care provider to “clear” the victim when the AED is ready to deliver the shock. The healthcare provider immediately pushes the shock button and resumes CPR immediately for another two minutes. The AED then analyzes the rhythm, charges, and prompts the healthcare provider to shock if necessary; the machine then prompts the resumption of CPR following the shock if necessary. This sequence repeats until the victim begins to move or regains consciousness or others arrive to take over the victim’s care. When a manual defibrillator is used, the first provider continues CPR for two minutes while a second provider applies defibrillator pads and charges the defibrillator. Once charged, CPR is paused to “clear” the patient. The shock is delivered once the patient is cleared and CPR is resumed for two minutes. The cycle is repeated beginning with a rhythm check.
- Waveform and Energies – For biphasic defibrillators, the manufacturer’s recommendations regarding effective energy doses should be followed for terminating VF (Class I). If the provider is unaware of the manufacturer’s suggested effective dose range, a maximum dose should be delivered (Class IIB). Subsequent energy levels delivered should be equivalent or higher. For monophasic defibrillators, a dose of 360J should be used for all shocks. A recurring VF after cardioversion should be shocked with the same dose that successfully terminated that VF initially.
- Multimodal Defibrillators: Automatic vs. manual modes – Multimodal defibrillators operating in manual mode reduce interruptions in CPR; however, there is the risk of an increase in the frequency of shocks delivered inappropriately.18-19 It remains to be seen whether the manual mode should be prioritized over the automatic mode (Class IIb).
- CPR before defibrillation: In cardiac arrest patients with VF or pulseless VT, the myocardium is deficient in oxygen and necessary nutrients. Two minutes of high-quality CPR can replenish these depleted resources. High-quality CPR can also help to reduce excess fluid volume in the right ventricle. This significantly increases the likelihood of the return of a perfusing rhythm after defibrillation.20 However, the value of intentionally delaying defibrillation in favor of CPR is not entirely clear.21-24
- VF Waveform Analysis to Predict Defibrillation Success – it is possible to predict the likelihood of successful defibrillation from VF waveforms; however, its predictive value in guiding rhythm-based management is less than certain (Class IIb).20, 25-28
- Drug therapy in VF/pulseless VT – Vasopressors (i.e. epinephrine) increase myocardial blood flow in CPR, thus increasing the chance of ROSC. The effect of vasopressors peak at one to two minutes after intravenous or intraosseous injection. The administration of vasopressors is performed right after a shock that fails to produce a perfusing rhythm. If a vasopressor is given after a shock that successfully generated a perfusing rhythm, it can have a detrimental effect on cardiovascular stability. Physiologic monitoring during CPR is essential to avoid this (quantitative waveform capnography, intra-arterial pressure monitoring, continuous venous oxygen saturation).29-39 Amiodarone is the first-line antiarrhythmic agent of choice that has been clinically proven to improve the chance of successful conversion to a perfusing rhythm. It can be considered if the patient in VF/pulseless VT is not responsive to CPR and defibrillation (Class IIb).
- Treating reversible causes of VF/pulseless VT – The health care provider should know the H’s and T’s mnemonic by heart (see Table 1). This is fundamental to the overall survival of the patient in cardiac arrest, as one (or possibly more than one) of these reversible problems may either have caused cardiac arrest or is preventing successful resuscitation.
- ROSC after VF/pulseless VT – Once the patient has achieved ROSC, post-cardiac arrest care should be started (this is will be discussed in future articles).
- PEA/asystole – PEA and asystole are NON-SHOCKABLE rhythms. In rhythms such as these, CPR is resumed immediately with rhythm checks every 2 minutes. Once a rhythm check reveals an organized rhythm, a pulse check is then performed. If there is a pulse, post-cardiac arrest care is given to the patient. If there is no pulse, CPR is continued for another 2 minutes, followed by a rhythm check. Monitoring of CPR quality is essential.
- Drug therapy for PEA/asystole – Vasopressors are given to increase myocardial blood flow and cerebral blood flow during CPR with the goal of achieving ROSC. Atropine is no longer used in the PEA/Asystole algorithm as it has been proven that this drug does not have any therapeutic effect in these rhythms.
- Treating potentially reversible causes of PEA/asystole (Table 1) – PEA (pulseless electrical activity, once called electromechanical dissociation) is often caused by hypoxemia, severe hypovolemia, pulmonary embolism and pneumothorax; appropriate treatment may reverse PEA. For example, in the case of hypoxemia, placement of an advanced airway should be prioritized. In volume loss or sepsis, intravenous or intraosseous administration of a crystalloid will benefit the patient. When blood loss has occurred in sufficient volume, a blood transfusion may reverse PEA. Fibrinolytic therapy may be considered in suspected arrest secondary to pulmonary embolism, while needle decompression should be performed in patients with tension pneumothorax.
- ROSC after PEA or asystole – Again, once the patient has achieved ROSC, post-cardiac arrest care should be started.
The way in which healthcare providers execute rhythm-based management will determine in part the outcome of the patient in cardiac arrest. In reality, there are far more unsuccessful resuscitative attempts than successful ones. Research continues to determine the best and most appropriate treatment modalities for victims of cardiac arrest.
Table 1. H’s and T’s
| H’s | T’s |
|---|---|
| Hypovolemia | Tension pneumothorax |
| Hypoxia | Tamponade, cardiac |
| Hydrogen ion (acidosis) | Toxins |
| Hypo/hyperkalemia | Thrombosis, pulmonary |
| Hypothermia | Thrombosis, coronary |
References:
- Chan PS, Krumholz HM, Nichol G, Nallamothu BK. Delayed time to defibrillation after in-hospital cardiac arrest. N Engl J Med. 2008; 358:9 –17
- Valenzuela TD, Roe DJ, Nichol G, Clark LL, Spaite DW, Hardman RG. Outcomes of rapid defibrillation by security officers after cardiac arrest in casinos. N Engl J Med. 2000;343:1206 –1209.
- The Public Access Defibrillation Trial Investigators. Public-access defibrillation and survival after out-of-hospital cardiac arrest. N Engl J Med. 2004;351:637– 646.
- Christenson J, Andrusiek D, Everson-Stewart S, Kudenchuk P, Hostler D, Powell J, Callaway CW, Bishop D, Vaillancourt C, Davis D, Aufderheide TP, Idris A, Stouffer JA, Stiell I, Berg R. Chest compression fraction determines survival in patients with out-of-hospital ventricular fibrillation. Circulation. 2009;120:1241–1247.
- Garza AG, Gratton MC, Salomone JA, Lindholm D, McElroy J, Archer R. Improved patient survival using a modified resuscitation protocol for out-of-hospital cardiac arrest. Circulation. 2009;119:2597–2605.
- Bobrow BJ, Clark LL, Ewy GA, Chikani V, Sanders AB, Berg RA, Richman PB, Kern KB. Minimally interrupted cardiac resuscitation by emergency medical services for out-of-hospital cardiac arrest. JAMA. 2008;299:1158 –1165.
- Kellum MJ, Kennedy KW, Barney R, Keilhauer FA, Bellino M, Zuercher M, Ewy GA. Cardiocerebral resuscitation improves neurologically intact survival of patients with out-of-hospital cardiac arrest. Ann Emerg Med. 2008;52:244 –252.
- Sayre MR, Cantrell SA, White LJ, Hiestand BC, Keseg DP, Koser S. Impact of the 2005 American Heart Association cardiopulmonary resuscitation and emergency cardiovascular care guidelines on out-of-hospital cardiac arrest survival. Prehosp Emerg Care. 2009;13:469–477.
- Steinmetz J, Barnung S, Nielsen SL, Risom M, Rasmussen LS. Improved survival after an out-of-hospital cardiac arrest using new guidelines. Acta Anaesthesiol Scand. 2008;52:908 –913.
- Aufderheide TP, Yannopoulos D, Lick CJ, Myers B, Romig LA, Stothert JC, Barnard J, Vartanian L, Pilgrim AJ, Benditt DG. Implementing the 2005 American Heart Association guidelines improves outcomes after out-of-hospital cardiac arrest. Heart Rhythm. 2010.
- Wong ML, Carey S, Mader TJ, Wang HE. Time to invasive airway placement and resuscitation outcomes after inhospital cardiopulmonary arrest. Resuscitation. 2010;81:182–186.
- Jennings PA, Cameron P, Walker T, Bernard S, Smith K. Out-of-hospital cardiac arrest in Victoria: rural and urban outcomes. Med J Aust. 2006;185:135–139.
- Kudenchuk PJ, Cobb LA, Copass MK, Cummins RO, Doherty AM, Fahrenbruch CE, Hallstrom AP, Murray WA, Olsufka M, Walsh T. Amiodarone for resuscitation after out-of-hospital cardiac arrest due to ventricular fibrillation. N Engl J Med. 1999;341:871– 878.
- Vandycke C, Martens P. High dose versus standard dose epinephrine in cardiac arrest—a meta-analysis. Resuscitation. 2000;45:161–166.
- Dorian P, Cass D, Schwartz B, Cooper R, Gelaznikas R, Barr A. Amiodarone as compared with lidocaine for shock-resistant ventricular fibrillation. N Engl J Med. 2002;346:884–890.
- Stiell IG, Wells GA, Field B, Spaite DW, Nesbitt LP, De Maio VJ, Nichol G, Cousineau D, Blackburn J, Munkley D, Luinstra-Toohey L, Campeau T, Dagnone E, Lyver M. Advanced cardiac life support in out-of-hospital cardiac arrest. N Engl J Med. 2004;351:647– 656.
- Olasveengen TM, Sunde K, Brunborg C, Thowsen J, Steen PA, Wik L. Intravenous drug administration during out-of-hospital cardiac arrest: a randomized trial. JAMA. 2009;302:2222–2229.
- Pytte M, Pedersen TE, Ottem J, Rokvam AS, Sunde K. Comparison of hands-off time during CPR with manual and semi-automatic defibrillation in a manikin model. Resuscitation. 2007;73:131–136.
- Kramer-Johansen J, Edelson DP, Abella BS, Becker LB,Wik L, Steen PA. Pauses in chest compression and inappropriate shocks: a comparison of manual and semi-automatic defibrillation attempts. Resuscitation. 2007;73:212–220.
- Eftestol T, Wik L, Sunde K, Steen PA. Effects of cardiopulmonary resuscitation on predictors of ventricular fibrillation defibrillation success during out-of-hospital cardiac arrest. Circulation. 2004;110: 10–15.
- Wik L, Hansen TB, Fylling F, Steen T, Vaagenes P, Auestad BH, Steen PA. Delaying defibrillation to give basic cardiopulmonary resuscitation to patients with out-of-hospital ventricular fibrillation: a randomized trial. JAMA. 2003;289:1389 –1395.
- Cobb LA, Fahrenbruch CE, Walsh TR, Copass MK, Olsufka M, Breskin M, Hallstrom AP. Influence of cardiopulmonary resuscitation prior to defibrillation in patients with out-of-hospital ventricular fibrillation. JAMA. 1999;281:1182–1188.
- Baker PW, Conway J, Cotton C, Ashby DT, Smyth J, Woodman RJ, Grantham H. Defibrillation or cardiopulmonary resuscitation first for patients with out-of-hospital cardiac arrests found by paramedics to be in ventricular fibrillation? A randomised control trial. Resuscitation. 2008; 79:424–431.
- Jacobs IG, Finn JC, Oxer HF, Jelinek GA. CPR before defibrillation in out-of-hospital cardiac arrest: a randomized trial. Emerg Med Australas. 2005;17:39–45.
- Box MS, Watson JN, Addison PS, Clegg GR, Robertson CE. Shock outcome prediction before and after CPR: a comparative study of manual and automated active compression-decompression CPR. Resuscitation. 2008;78:265–274.
- Brown CG, Dzwonczyk R, Martin DR. Physiologic measurement of the ventricular fibrillation ECG signal: estimating the duration of ventricular fibrillation. Ann Emerg Med. 1993;22:70 –74.
- Callaway CW, Sherman LD, Mosesso VN Jr, Dietrich TJ, Holt E, Clarkson MC. Scaling exponent predicts defibrillation success for out-of-hospital ventricular fibrillation cardiac arrest. Circulation. 2001;103: 1656–1661.
- Eftestol T, Sunde K, Aase SO, Husoy JH, Steen PA. Predicting outcome of defibrillation by spectral characterization and nonparametric classification of ventricular fibrillation in patients with out-of-hospital cardiac arrest. Circulation. 2000;102:1523–1529.
- Callaham M, Barton C. Prediction of outcome of cardiopulmonary resuscitation from end-tidal carbon dioxide concentration. Crit Care Med. 1990;18:358 –362.
- Paradis NA, Martin GB, Rivers EP, Goetting MG, Appleton TJ, Feingold M, Nowak RM. Coronary perfusion pressure and the return of spontaneous circulation in human cardiopulmonary resuscitation. JAMA. 1990;263:1106 –1113.
- Rivers EP, Martin GB, Smithline H, Rady MY, Schultz CH, Goetting MG, Appleton TJ, Nowak RM. The clinical implications of continuous central venous oxygen saturation during human CPR. Ann Emerg Med. 1992;21:1094 –1101.
- Cantineau JP, Lambert Y, Merckx P, Reynaud P, Porte F, Bertrand C, Duvaldestin P. End-tidal carbon dioxide during cardiopulmonary resuscitation in humans presenting mostly with asystole: a predictor of outcome. Crit Care Med. 1996;24:791–796.
- Grmec S, Kupnik D. Does the Mainz Emergency Evaluation Scoring (MEES) in combination with capnometry (MEESc) help in the prognosis of outcome from cardiopulmonary resuscitation in a pre-hospital setting? Resuscitation. 2003;58:89 –96.
- Grmec S, Lah K, Tusek-Bunc K. Difference in end-tidal CO2 between asphyxia cardiac arrest and ventricular fibrillation/pulseless ventricular tachycardia cardiac arrest in the prehospital setting. Crit Care. 2003;7: R139–R144.
- Grmec S, Klemen P. Does the end-tidal carbon dioxide (EtCO2) concentration have prognostic value during out-of-hospital cardiac arrest? Eur J Emerg Med. 2001;8:263–269.
- Kolar M, Krizmaric M, Klemen P, Grmec S. Partial pressure of end-tidal carbon dioxide successfully predicts cardiopulmonary resuscitation in the field: a prospective observational study. Crit Care. 2008;12:R115.
- Steedman DJ, Robertson CE. Measurement of end-tidal carbon dioxide concentration during cardiopulmonary resuscitation. Arch Emerg Med. 1990;7:129 –134.
- Grmec S, Mally S. Timeliness of administration of vasopressors in CPR. Crit Care. 2009;13:401.
- Pokorna M, Necas E, Kratochvil J, Skripsky R, Andrlik M, Franek O. A sudden increase in partial pressure end-tidal carbon dioxide (P(ET)CO(2)) at the moment of return of spontaneous circulation. J Emerg Med. 2009;38:614–621.
Recommended Articles

Adult Cardiac Arrest – Ventricular Fibrillation Video
Ready for another rapid algorithm review? Learn more about ventricular fibrillation and adult cardiac arrest.